Discover Your Career At SMC Ltd.
WHO WE ARE
A Strategic Manufacturing Partner for Healthcare Innovators
SMC Ltd. was developed around a commitment to its people and customers. This continued focus has helped us become a global leader in contract manufacturing for medical device, diagnostics and drug delivery companies.
With continued growth and success, we seek individuals who are interested in providing top-notch customer service and world-class innovation in manufacturing devices that improve and save the lives of patients throughout the world

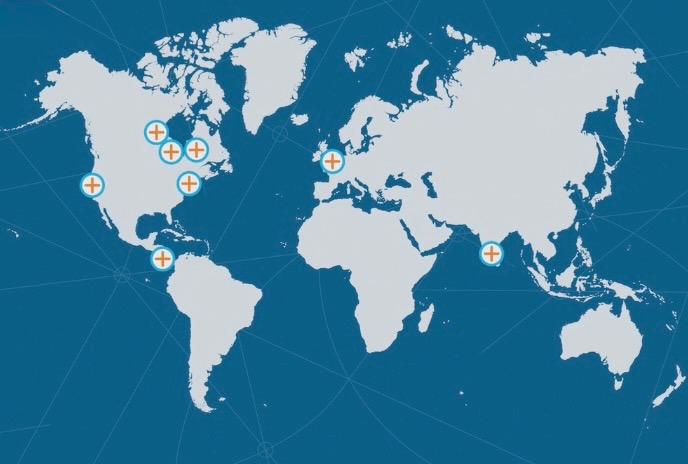
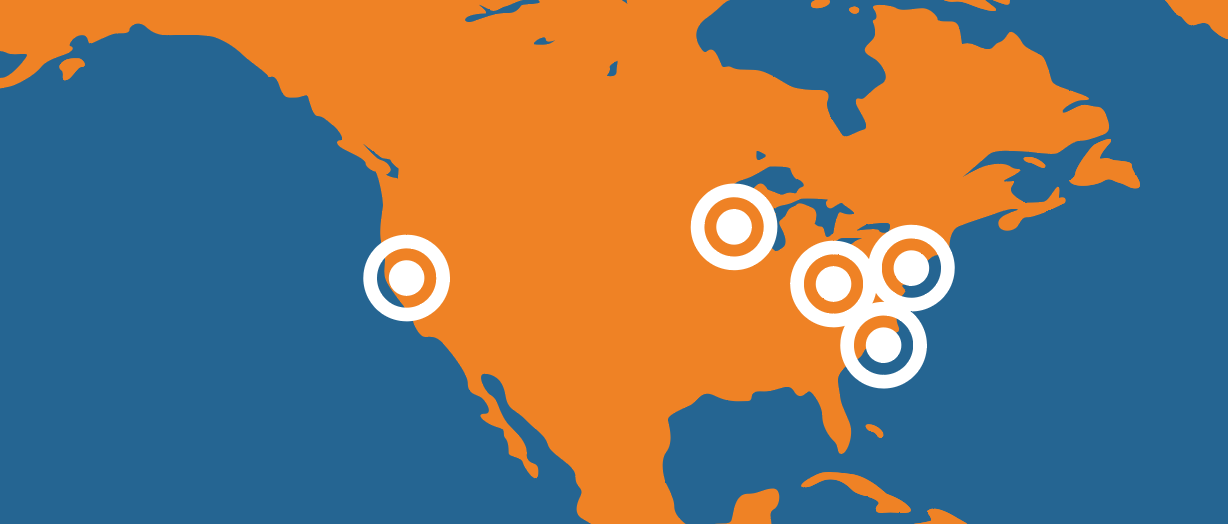
LOCATIONS
Select a Location Below to Search for Open Positions